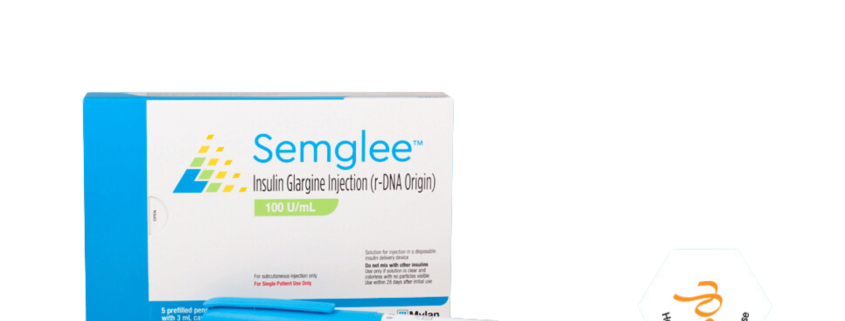
SEMGLEE

Generic Name: Insulin Glargine Injection (r-DNA Origin) 100 U/mL
Dosage Form: Injection
TG Name: Endocrine & Diabetes
1. What is SEMGLEE™?
SEMGLEE™ is a long-acting man-made insulin used to control high blood sugar in adults with diabetes mellitus.
2. How SEMGLEE™ works in our body?
Insulin is a hormone produced in the pancreas by cells called “beta cells”. We get most of our energy from blood glucose from food. Insulin helps the glucose in the blood get in to our cells, which they then use for energy. In someone with type 1 diabetes, the pancreas has stopped producing insulin and so it has to be replaced by insulin shots so their bodies can use the glucose from the food they eat. Someone with type 2 diabetes still produces some insulin but their bodies don’t respond to it enough and need diabetes pills or insulin shots so their bodies can use the glucose from the food they eat for energy.
SEMGLEE™ is a long-acting human insulin analog indicated to improve glycemic control in adults and pediatric patients with type 1 diabetes mellitus and in adults with type 2 diabetes mellitus.
3. What are the indications of SEMGLEE™?
- SEMGLEE™ is indicated to improve glycemic control in adults and pediatric patients with type 1 diabetes mellitus and in adults with type 2 diabetes mellitus
- SEMGLEE™ is not for use to treat diabetic ketoacidosis
- It is not known if SEMGLEE™ is safe and effective in children less than 6 years of age with type 1 diabetes
- It is not known if SEMGLEE™ is safe and effective in children with type 2 diabetes
4. What are the dosage & administration of SEMGLEE™?
General Dosing Instruction:
- Individualize and adjust the dosage of SEMGLEE™ based on the individual’s metabolic needs, blood glucose monitoring results and glycemic control goal
- Dosage adjustments may be needed with changes in physical activity, changes in meal patterns (i.e., macronutrient content or timing of food intake), during acute illness, or changes in renal or hepatic function. Dosage adjustments should only be made under medical supervision with appropriate glucose monitoring
Initiation of SEMGLEE™ Therapy:
Type 1 Diabetes
In patients with type 1 diabetes, SEMGLEE™ must be used concomitantly with short-acting insulin. The recommended starting dose of SEMGLEE™ in patients with type 1 diabetes should be approximately one-third of the total daily insulin requirements. Short-acting, pre-meal insulin should be used to satisfy the remainder of the daily insulin requirements.
Type 2 Diabetes
The recommended starting dose of SEMGLEE™ in patients with type 2 diabetes who are not currently treated with insulin is 0.2 units/kg or up to 10 units once daily. One may need to adjust the amount and timing of short-or rapid-acting insulins and dosages of other antidiabetic drugs.
Changing to SEMGLEE™ from Other Insulin Therapies:
- If changing patients from once-daily insulin glargine injection 300 units/mL to once-daily SEMGLEE™, the recommended initial SEMGLEE™ dose is 80% of the insulin glargine injection 300 units/mL dose that is being discontinued. This dose reduction will lower the likelihood of hypoglycemia
- If changing from a treatment regimen with an intermediate or long-acting insulin (other than an insulin glargine injection 100 units/mL) to a regimen with SEMGLEE™, a change in the dose of the basal insulin may be required and the amount and timing of the shorter-acting insulins and doses of other antidiabetic drugs may need to be adjusted
- In patients changing from once-daily NPH insulin to once daily dose of SEMGLEE™, the recommended initial SEMGLEE™ dose is the same as the dose of NPH that is being discontinued
- If changing patients from twice-daily NPH insulin to once-daily SEMGLEE™, the recommended initial SEMGLEE™ dosage is 80% of the total NPH dose that is being discontinued. This dosage reduction will lower the likelihood of hypoglycemia
5. What are the contraindications of SEMGLEE™?
- During episodes of hypoglycemia
- In patients with hypersensitivity to insulin glargine or one of its excipients
6. What are the adverse effects of SEMGLEE™?
- Hypoglycemia
- Medication Errors
- Hypersensitivity and allergic reactions
- Hypokalemia
7. What would be the storage condition of SEMGLEE™?
Unopened: Unopened SEMGLEE™ devices should be stored in a refrigerator, 36° to 46°F (2° to 8°C). Do not freeze. Discard if frozen. Discard after the expiration date.
Open (In-Use): The opened (in-use) SEMGLEE™ should NOT be refrigerated but should be kept at room temperature (below 86°F [30°C]) away from direct heat and light. The opened (in-use) SEMGLEE™ device must be discarded 28 days after being opened.